Tetrahydro-4H-Pyran-4-One
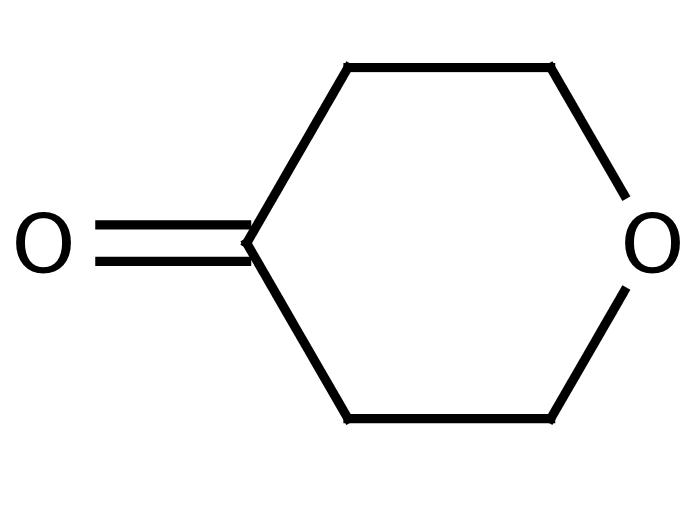
It is employed in the preparation of 4-methoxytetrahydropyran-4-yl protecting group, synthesis of symmetric tetra substituted methanes. The methyl enol ether is a useful protecting agent for alcohols, e.g. in nucleotide synthesis, with the advantage over 3,4-Dihydro-2H-pyran. It is also employed in a study of the enantioselective alpha-aminoxylation of ketones with nitrosobenzene and L-proline in an ionic liquid. It undergoes condensation reactions in the preparation of dipeptides and spiroimidazolones. It is also employed in wittig reactions for the synthesis of Penicillins and in a ring of vitamin D3.
Product Details
| Category |
Heterocycles & Building Blocks |
|---|---|
| Price | POA |
| CAS Number | 29943-42-8 |
| Formula | C5H8O2 |
| Molecular Weight | 100.12 |
| Purity | 99.0% |
| Appearance | colourless to light yellow liquid |
| Availability | In Stock |
| Storage Conditions | Room temperature |
Product Enquiry
Send us your enquiry for Tetrahydro-4H-Pyran-4-One. We offer custom pack sizes at special prices. We aim to respond to your enquiry within 24 hours.
We value your input so if you have suggestions regarding new applications for Tetrahydro-4H-Pyran-4-One email us and we will include your contribution on the website.



