Raltegravir
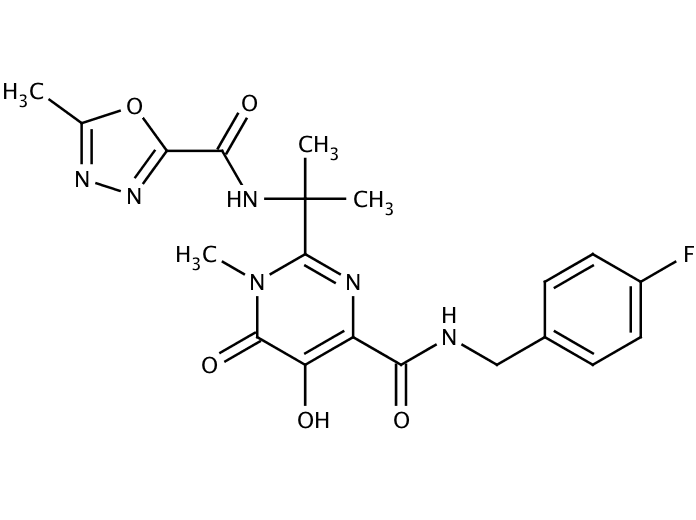
Raltegravir is an antiretroviral drug used to treat HIV infection. It received approval by the U.S. Food and Drug Administration (FDA) on 12 October 2007, the first of a new class of HIV drugs, the integrase inhibitors, to receive such approval. Raltegravir targets integrase, an HIV enzyme that integrates the viral genetic material into human chromosomes, a critical step in the pathogenesis of HIV. The drug is metabolized away via glucuronidation.
Product Details
| Category |
Cancer Research and Antivirals |
|---|---|
| Price | POA |
| CAS Number | 518048-05-0 |
| Formula | C20H21FN6O5 |
| Molecular Weight | 442.42 |
| Purity | 98.0% |
| Availability | In Stock |
| Storage Conditions | -18°C |
Product Enquiry
Send us your enquiry for Raltegravir. We offer custom pack sizes at special prices. We aim to respond to your enquiry within 24 hours.
We value your input so if you have suggestions regarding new applications for Raltegravir email us and we will include your contribution on the website.



