Ammonium Iron(II) Sulfate Hexahydrate
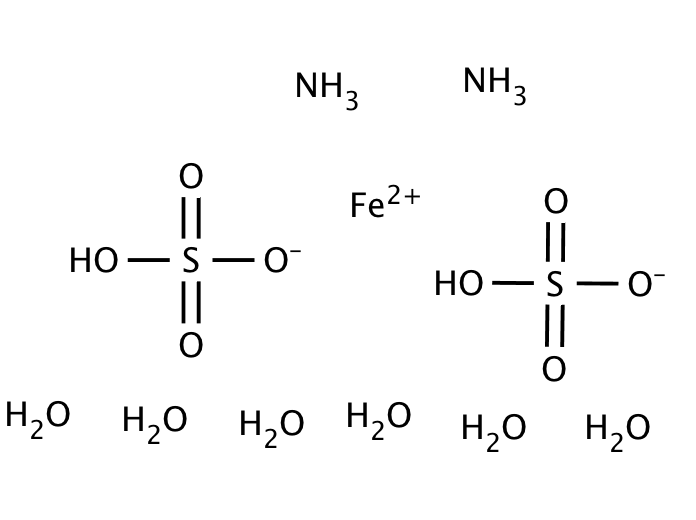
Ammonium iron(II) sulfate hexahydrate or Mohr’s Salt is often employed as an analytical standard, and has been used in a variety of other applications from nanomaterials to general redox reactions. This product is much less affected by oxygen in the air than iron(II) sulfate, making it more desirable for titration purposes, where iron(II) might be oxidized to iron(III). The oxidation of iron(II) to iron(III) is pH dependent, and the reaction occurs more rapidly at higher pH; Mohr’s Salt lowers the pH of solutions slightly, thereby preventing oxidation from occurring.
Product Details
| Category |
Laboratory reagents and chemicals |
|---|---|
| Price | POA |
| CAS Number | 7783-85-9 |
| Formula | (NH4)2Fe(SO4)2 · 6H2O |
| Molecular Weight | 392.14 |
| Purity | 99.0% |
| Appearance | crystalline or powder |
| Availability | In Stock |
| Storage Conditions | Room temperature |
Product Enquiry
Send us your enquiry for Ammonium Iron(II) Sulfate Hexahydrate. We offer custom pack sizes at special prices. We aim to respond to your enquiry within 24 hours.
We value your input so if you have suggestions regarding new applications for Ammonium Iron(II) Sulfate Hexahydrate email us and we will include your contribution on the website.



